I recently went to an event hosted by the Crohn’s and Colitis Foundation (CCF)’s Northern California chapter. The event was an educational symposium for patients and families with IBD, and I was impressed with the CCF organization as well as the amazing community of patients and caregivers. I want to thank Kayla Kraich for all of her hard work organizing such a nice and welcoming event.
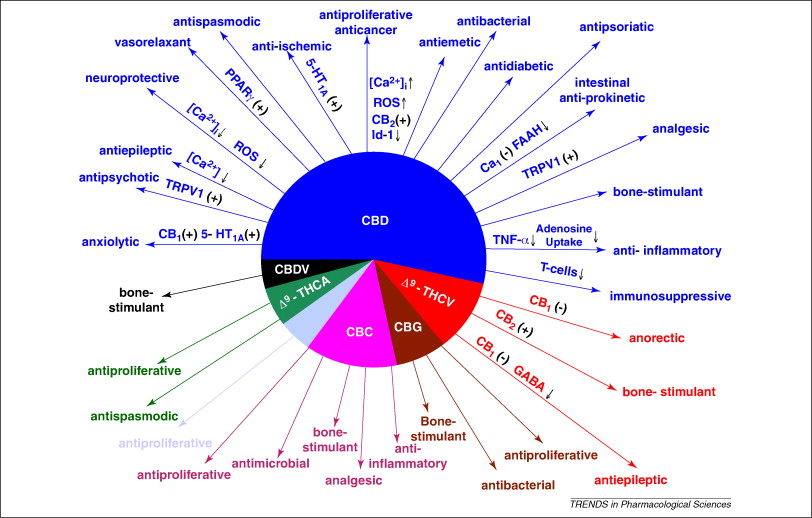
I went to the CCF event to learn more about IBD, and specifically biologic treatments for IBD, their benefits, as well as their limitations. Our startup is developing a new small-molecule treatment of IBD, so I would like to stay on top of the standard of care and see what else is going on in the field.
I grabbed a cup of coffee, sat down, and waited for the talks to start. The people sitting around me all looked like happy and healthy adults leading vibrant lives. The guy in front of me looked about my age, well dressed, clean-cut, and maybe a touch sciencey. I thought, hey, maybe he is another researcher? The first talk focused on biologics for IBD, when they are indicated, pros and cons, etc. At the end of the talk the MD invited up a panel of patients to talk about their experience with biologics. The man in front of me stood up and joined the patient panel. When he shared his story of battling IBD since he was 15, it was eye-opening. I have heard that IBD is one of the most disruptive diseases that can be largely hidden from the outside world.
At the first break, the woman sitting next to me introduced herself. Right after her name, she launched into her history with ulcerative colitis, her multiple surgeries, and how she ultimately tamed her disease with a balance of diet and medications. At the end of her quick life story, she then said, “what about you Brandon?”
I have to admit that I felt like a poser. I don’t have IBD. I haven’t had multiple surgeries. I haven’t seen numerous doctors, received infusions of antibodies, worried about infections from a suppressed immune system. I haven’t struggled to knock down GI flares with steroids, and then dealing with their crazy side effects. Nor have I endured the never-ending visits to the bathroom (a metric for ulcerative colitis activity is the number of daily bowel movements, with the highest tier being anything over 10 times a day).
I told her that I’m just a researcher, but we are working on IBD and have a new drug that we are rapidly advancing to the clinic. She put me at ease with her genuine appreciation for my research, and although I felt insignificant and like I didn’t belong, she welcomed me to the event and wished me the best of luck in our work.
At this point, I may have drank a couple cups of coffee trying to remain alert and sharp during the morning talks, so I made my first trip to the men’s room.
What I encountered in the men’s room was subtle at first, but after recounting the experience and putting it into context with the diseases of the colon, it started to make sense. On my first trip to the men’s room I noticed a number of people coming and going, but the notable interaction was with someone who introduced himself while walking from the urinal to the sinks. The man was very extroverted, and the tone and content of the conversation could have taken place in any setting. Only this time, it was in the men’s room. And that level of forward communication in the men’s room seemed a little odd to me.
Back to the talks, the next speaker gave a nice overview of IBD (the MD and I overlapped as undergrads at UC Davis- go Ags!). After this second talk, my bladder was losing the battle to the coffee and water I was drinking, so I made my second trip to the men’s room. This trip, there was another very genuine introduction and discussion that started in the bathroom. I chatted, and eventually made my way back to the conference room, and didn’t think much of it.
After the talks I headed home, and during the drive I reflected on the great talks and on my interactions with the amazing members of the CCF community. And of course those very genuine interactions that I had in the men’s room…
And then it hit me.
For a patient population that uses the restroom so much, the CCF community is very, very comfortable in the bathroom. Especially at an event filled with fellow patients, doctors, and caregivers, and when it is hard to tell who has what disease, everyone wearing a CCF nametag must seem like an ally. I laughed out loud when I put it together. And then the realization actually felt heavy on my face and my chest. There was a lifelong struggle that was underneath the interactions with the people in the bathroom. I have now attended two CCF events, at which the first item of business was telling everyone the location of the bathrooms. My gastrointestinal health is something I have always taken for granted, but I am now looking at it with a new perspective.
The more that I learn about Crohn’s and Colitis, it just kills me to think of what IBD sufferers are going through. As we are so preoccupied with the pharmacology and interactions of our drug compounds in lab, we don’t often think about of these complications that IBD patients suffer daily. I am extremely grateful for how welcoming and open the CCF community has been with me. It leaves me feeling hungry for our upcoming clinical trials, and it reaffirms that IBD is a worthy cause that I am grateful to be working on.
-BZ